38th ACS National Medicinal Chemistry Symposium
Join Optibrium’s Chris Khoury at the 38th NMCS meeting in Seattle, 23-26 June

Join Optibrium’s Chris Khoury at the 38th NMCS meeting in Seattle, 23-26 June

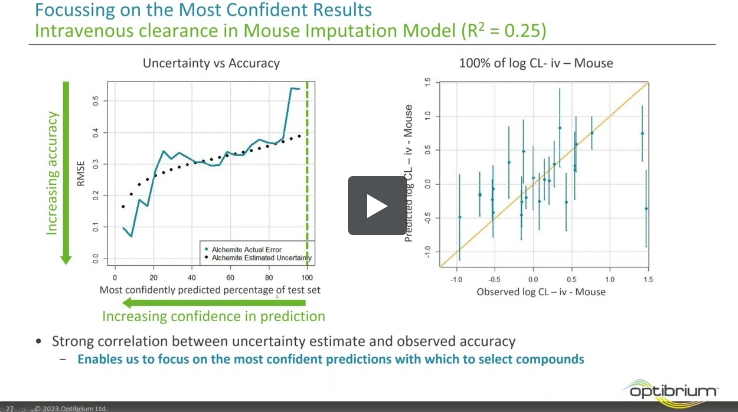
If you’re using predictive models in your molecule design and optimisation, an accurate uncertainty estimate can be just as important…

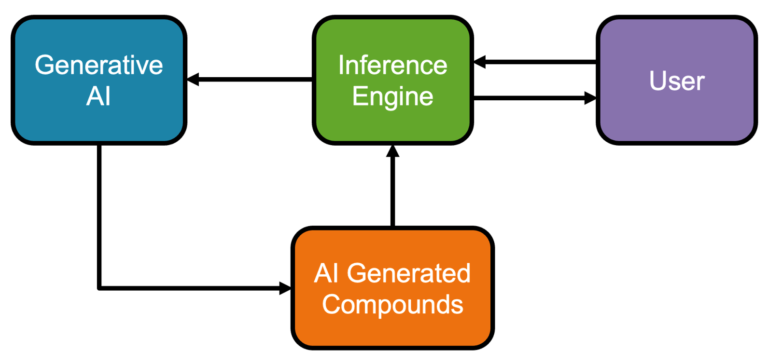
Pairing AI with human expertise We present a novel AI compound optimisation system, designed to include human oversight as a…
Introduction The emergence of resistance and increased stringency of regulatory requirements have created a need for new agrochemicals. The long…
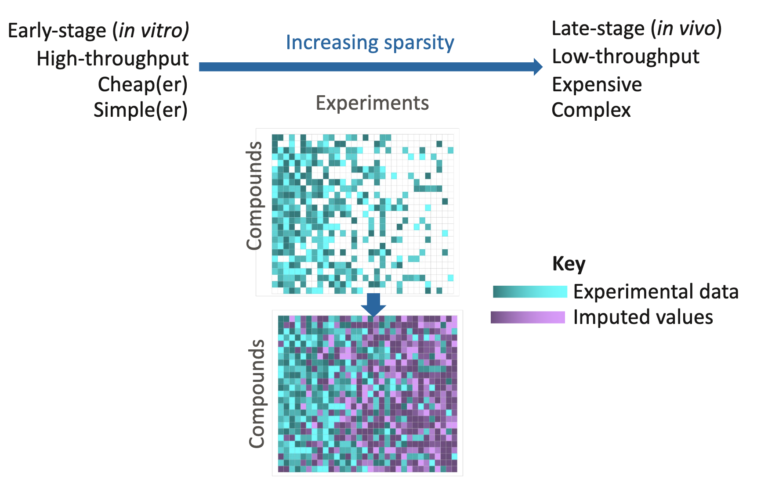
Recent years have seen a remarkable rise in the number and scope of artificial intelligence and machine learning (especially deep…


AI has the potential to transform discovery. However, to ensure real impact, there are several practicalities that organisations must consider…

Now, watch Matt Segall, PhD, CEO at Optibrium, as he introduces a real world case study where we applied deep learning to guide a project, in which potential compounds were displaying good activity profiles but the team wanted to improve their PK profile to achieve better efficacy.
In the face of growing agrochemical resistance and increasingly stringent regulatory requirements, how can artificial intelligence (AI) be harnessed to help lower the costs, failure rates and timelines associated with current agrochemical development cycles?
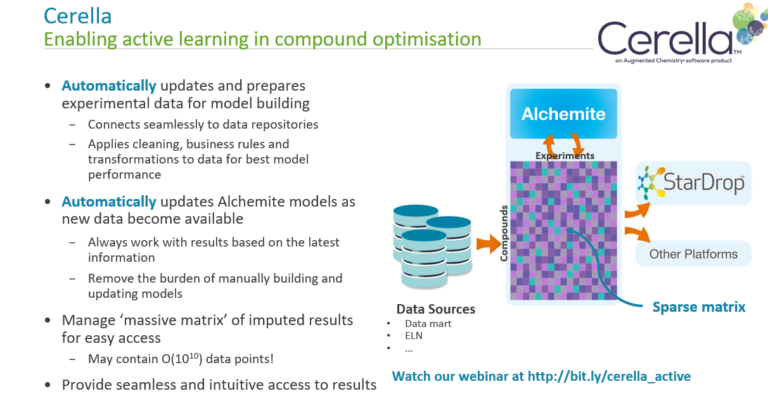
In this webinar, learn about Cerella’s unique AI methods, see examples of its successful application throughout the drug discovery process and watch a demonstration of how CDD Vault and Cerella connect to seamlessly integrate with your workflows.

In this webinar, we examine the effective use of QSAR modelling in drug discovery and discuss a variety of pain points for medicinal chemists in knowing when a model can be trusted and how to avoid common pitfalls.
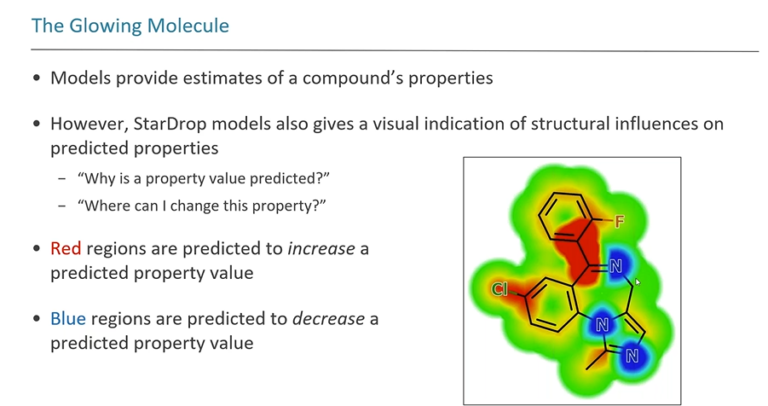
In this webinar, Jeff Blaney (Senior Director of Discovery Chemistry, Genentech), Darren Green (Head of Cheminformatics & Data Science, GlaxoSmithKline), Julian Levell (Head of Discovery, New Equilibrium Biosciences), Matthew Segall (CEO, Optibrium) discuss the state of AI in early drug discovery from hit to preclinical candidate and share their experiences with and expectations of AI, including predictive modelling, synthesis prediction, and generative chemistry. Hear about the successes of AI drug discovery and an outlook on what AI needs to achieve to really transform the industry.

In this webinar, we explore the highlights of collaborative project results that demonstrate how every phase of the drug discovery process can be radically improved by applying proven AI technology. Providing scientists with insights on which to base decisions can identify valuable new opportunities and reduce the time and cost of AI drug discovery cycles.
We review case studies from collaborations with Constellation Pharmaceuticals, AstraZeneca, Genentech, the University of Dundee and Takeda Pharmaceuticals to validate the impact of applying AI to experimental data and illustrate dramatic improvements to their project outcomes.
Join Samar Mahmoud and Matt Segall for this fascinating deep dive into the revolution that AI is bringing to the challenges of sparse and noisy drug discovery data.
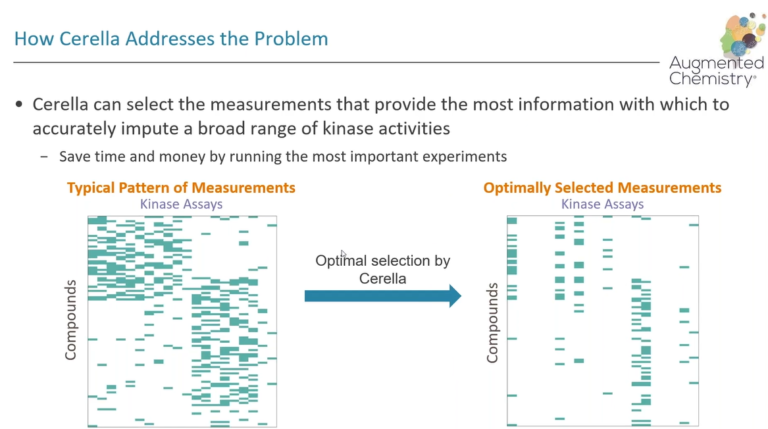
This article is a collaboration with Intellegens, the University of Cambridge and AstraZeneca. It provides a proof-of-concept study in which Cerella™ is used to predict rat in vivo pharmacokinetic (PK) parameters and concentration–time PK profiles.
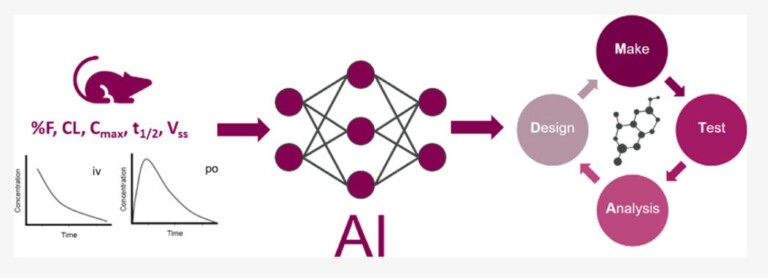
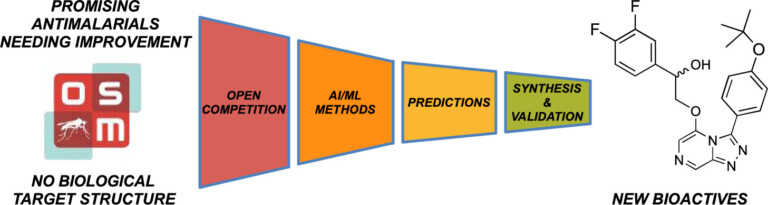
In this study, we identified a new antimalarial with an unusual structure – the only compound in the competition to be proven active, opening up new chemistry for exploration.
In this article, the team demonstrates the application of Alchemite™, a deep learning imputation method which underpins our Cerella™ technology, to physicochemical and sensory data.
In this webinar, we discuss Alchemite™, a novel deep learning approach, and its application to optimising kinase profiling programmes. The…

OA paper outlining the practical applications of deep imputation on large-scale drug discovery data. It compares deep learning to traditional QSAR methods.

In this webinar, we described the generation and validation of a ‘global’ model using deep learning imputation on a data set of 300,000 compounds and 500 experimental endpoints, targeting global health indications.
We demonstrated how this global model can be applied to individual optimisation projects, offering improved compounds design performance over ‘local’ project-specific models by learning across a broad chemical diversity.