Honolul, Hawaii
September 15 – 18, 2024
If you are heading to Honolulu for this years joint ISSX and JSSX meetings then you can hear more about how to ‘Address common metabolism problems in drug discovery with In Silico methods’. Join Chris Khoury and Daniel Barr, PhD to learn about how Optibrium’s software, tailored to DMPK scientists, is changing the landscape of accurate metabolism insight generation.
Come and meet the team to learn about how Optibrium’s range of drug discovery software can increase the speed, efficiency, and productivity of your chemistry discovery programs.
Abstract:
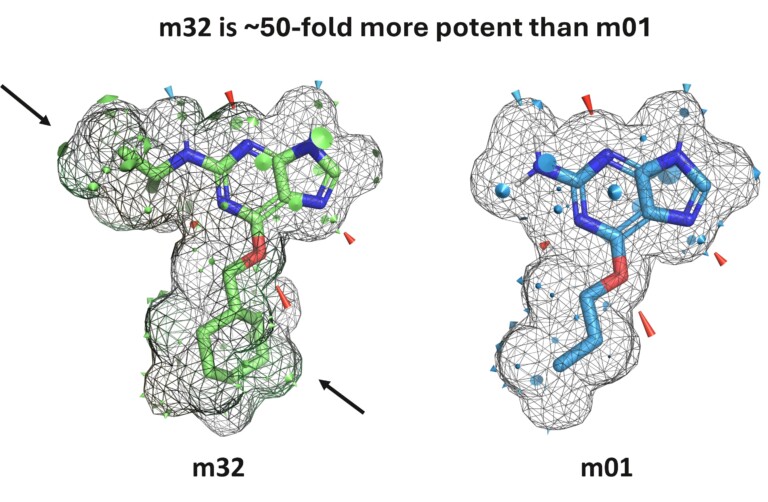
Authors: Daniel A. Barr, Mario Öeren, Peter A. Hunt, Jonathan D. Tyzack, Tomáš Chrien, Tamsin E. Mansley, and Matthew D. Segall
In silico metabolism prediction can address critical questions to guide lead optimization. Using several case studies, we demonstrate the application of these models to address design challenges involving metabolic (in)stability, the formation of reactive and/or toxic intermediates, and to mitigate the risk of genetic polymorphisms and drug-drug interactions.
In addition, we illustrate how these models can inform the selection of in vitro and in vivo pre-clinical experiments to avoid surprises in late-stage trials. Furthermore, accurate predictions of metabolite profiles early in the discovery process provide essential guidance for drug design. Optibrium’s mechanistic metabolism models cover metabolism by P450, AOX, FMO, UGT, and SULT enzymes1-5. By combining these models, metabolic pathway analysis proposes the most likely metabolites with greater precision than other methods, assisting in metabolite identification studies and enabling potentially active, reactive, or toxic metabolites to be identified6.
References:
- Mario Öeren, Peter J. Walton, James Suri, David J. Ponting, Peter A. Hunt and Matthew D. Segall, (2022) J. Med. Chem. 65(20) pp. 1406-1408
- Mario Öeren, Sylvia C. Kaempf, David J. Ponting, Peter A. Hunt and Matthew D. Segall, (2023) J. Chem. Inf. Model. 63(11) pp. 3340-3349
- Mario Öeren, Peter J. Walton, Peter A. Hunt, David J. Ponting and Matthew D. Segall, (2021) J. Comput.-Aided Mol. Des. 35(4) pp. 541-555
- Jonathan D. Tyzack, Peter A. Hunt and Matthew D. Segall, (2016) J. Chem. Inf. Model. 56(1) pp. 2180-2193
- Peter A. Hunt, Matthew D. Segall & Jonathan D. Tyzack, (2018) J. Comput.-Aided Mol. Des., 32 pp. 537-546
- Mario Öeren, Peter A. Hunt, Charlotte E. Wharrick, Hamed Tabatabaei Ghomi and Matthew D. Segall, (2023) Xenobiotica DOI: 10.1080/00498254.2023.2284251
Related content from across the site
Optibrium Introduces Graphical Interface for QuanSA to Enhance Ligand-Based Affinity Predictions
New user interface provides visual insights to guide compound optimisation CAMBRIDGE, UK, 24 March 2026 – Optibrium, a leading developer of software and AI solutions for molecular design, today…
Optibrium Appoints Dr Nathan Brown as Director of Science
Newly created role underscores commitment to customer-driven research and scientific excellence CAMBRIDGE, UK, 13 January 2026 – Optibrium, a leading developer of software and AI…

Optibrium Delivers Real-Time Collaboration in its Comprehensive Molecular Design Platform with StarDrop 8
Latest release accelerates hit-to-candidate progression by keeping teams aligned, eliminating inefficiencies and enabling better prioritisation decisions CAMBRIDGE, UK, 04 November…


